Introduction
Regulatory T cells (Tregs) blunt anti-tumor responses of major immune effector cells including CD8 T cells and natural killer (NK) cells. Tregs constitutively express CTLA-4 and thus the use of ipilimumab, a CTLA-4 blocker can potentially overcome the Treg-mediated immune suppression. We (and others) have shown that a brief activation of conventional NK cells with the cytokine combination IL-12, IL-15, and IL-18 generates cytokine-induced memory-like (CIML) NK cells which exhibit enhanced anti-tumor responses, have a longer half-life (up to 90 vs ~12 days of conventional NK cells), and show promising activity in several early phase clinical trials. N-803 is an IL-15 super-agonist with a long half-life that preferentially activates and expands NK and CD8+ T cells without affecting Tregs, making it an ideal cytokine for combining with adoptive immunotherapy. In the current study, we hypothesized the use of ipilimumab (IPI) will abrogate Treg-mediated inhibition and thus allow enhanced proliferation, activation, and anti-tumor responses of the adoptively transferred CIML NK cells.To address this, we conducted a phase 1 trial of HLA-haploidentical CIML NK cells in combination with IPI and N-803 in patients with advanced head and neck cancer (HNC). Using flow cytometry and CITE-Seq on the banked peripheral blood samples, we performed an in-depth characterization of NK cells expanded following IPI pre-exposure and compared it to those who did not receive IPI.
Methods
We enrolled 10 patients with refractory HNC regardless of human papillomavirus (HPV) status who had prior platinum and immunotherapy (NCT04290546). All patients received lymphodepletion with fludarabine (25 mg/m 2 x 5 days) and cyclophosphamide (60 mg/kg x 2 days) during days -6 to -2 prior to haploidentical CIML NK cell infusion on day 0 (5-10 x 10 6 cells/kg=dose level 0) followed by N-803 (15 mcg/kg subcutaneously) starting on day +1 every 21-days for 4-doses. In cohort 2, patients received IPI (3 mg/kg) on day -7, no ipilimumab was given to the first 6 patients treated on the lead-in safety cohort 1. Flow cytometry using a custom NK cell panel was performed on samples collected on days +7, +14, +21, +28, +42, and +60 after CIML NK cell infusion. FlowSOM clustering was performed on NK cell markers. UMAP clustering was applied to CITE sequencing data acquired from samples collected on day +7 and day +28, and well-defined markers genes for each cluster were used to identify cell populations. NK cell function (cytotoxicity, IFNg, and CD107a) was assessed using flow-cytometry based assays.
Results
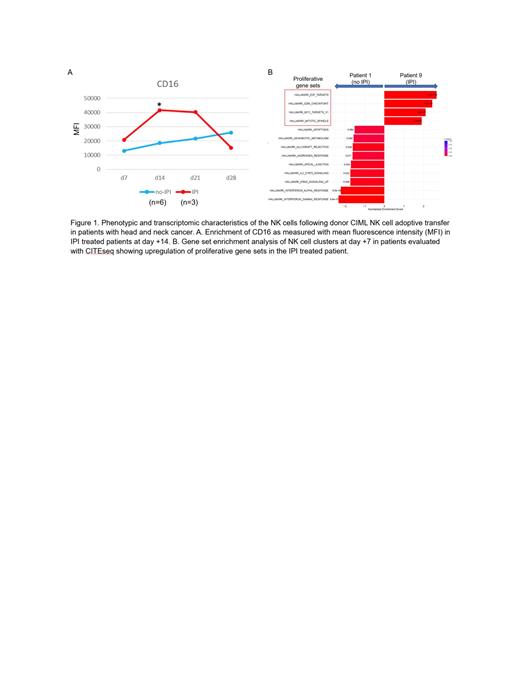
The median age of the patients was 58, 90% were men and 80% had oropharyngeal primaries. Mild CRS (grade 2 or lower) was observed in 60% of patients. No neurotoxicity or graft-versus-host disease was observed. Maximal expansion of NK cells occurred on day +7 following CIML NK cell infusion in 9/10 of patients. The single patient without NK cell expansion was found to have a donor-specific anti-HLA antibody. Day +7 NK cells exhibited upregulation of IFNg expression following cytokine restimulation as compared to resting NK cells. Tumor regression was observed in 50% of treated patients at day +30 based on RECIST v1.1 criteria. Patients who had tumor regression had an expansion of CD56 dimCD16 + NK cell clusters by day +14. IPI treated patients were noted to have a relative enrichment of CD16 + NK cell metaclusters at day +14 compared to no IPI patients (Figure 1A). At the time of maximal NK cell expansion, CITEseq defined NK cell clusters were dominant in both IPI and no IPI patients, but the proliferative gene sets were enriched in the IPI treated patient (Figure 1B). IPI exposure had no effect on the distribution of regulatory T cells or CD8 + T cell populations at day +28 compared to the no IPI patients.
Conclusions
The use of donor-derived CIML NK cells is safe and associated with tumor regression in patients with advanced HNC. The use of IPI was associated with the preferential expansion of CD16 + NK cell clusters that had enrichment of proliferative gene sets. Tumor regression was associated with CD56 dimCD16 + NK cell expansion. Further work is required to elucidate the mechanism by which IPI exposure is associated with the expansion of specific subsets of CIML NK cells.
Disclosures
Sade-Feldman:Bristol Myer-Squibb: Research Funding; Galvezibne Therapeutics: Consultancy. Vergara-Cadavid:Senti Bio: Current Employment. Koreth:Miltenyi Biotec: Research Funding; BMS: Research Funding; Clinigen Labs: Consultancy, Research Funding; Amgen: Research Funding; Tr1x: Consultancy; Biolojic Design: Consultancy; Cue Biopharma: Consultancy; Gentibio: Consultancy; Equillium: Consultancy; Mallinckrodt: Membership on an entity's Board of Directors or advisory committees; Regeneron: Research Funding; Cugene: Membership on an entity's Board of Directors or advisory committees; Equillium: Research Funding. Ritz:Kite/Gilead: Research Funding; Equillium: Research Funding; Garuda Therapeutics: Consultancy, Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees; Oncternal: Research Funding; TScan Therapeutics: Consultancy, Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees; LifeVault Bio: Consultancy, Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Smart Immune: Consultancy, Membership on an entity's Board of Directors or advisory committees; Avrobio: Consultancy, Membership on an entity's Board of Directors or advisory committees; Clade Therapeutics: Consultancy, Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees; Akron Biotech: Consultancy, Membership on an entity's Board of Directors or advisory committees. Nikiforow:Sobi: Other: Participation in ad hoc advisory board; Kite/Gilead: Other: Participation in ad hoc advisory board; Iovance: Other: Participation in ad hoc advisory board; GlaxoSmithKline: Other: Participation in ad hoc advisory board; A2 Bio: Other: Participation in ad hoc advisory board. Soiffer:Neovii: Consultancy; Juno Therapeutics/ BMS/Celgene USA: Other: Data Safety Monitoring Board; Smart Immune: Consultancy; Bluesphere Bio: Consultancy; Vor Bipharma: Consultancy; Jasper: Consultancy; NMPD - Be the Match, USA: Membership on an entity's Board of Directors or advisory committees; Astellas: Consultancy. Hanna:Bicara: Research Funding; BMS: Honoraria, Research Funding; Exicure: Honoraria, Research Funding; Gateway for Cancer Research: Research Funding; GSK: Research Funding; ImmunityBio: Research Funding; Kite: Research Funding; KSQ: Honoraria, Research Funding; Kura: Honoraria, Research Funding; Regeneron: Research Funding; Repertoire: Research Funding; Sanofi Genzyme: Honoraria, Research Funding; Secura Bio: Research Funding; Bicara: Honoraria; Bio-Rad: Honoraria; Boxer Capital: Honoraria; Coherus: Honoraria; General Catalyst: Honoraria; Massachusetts Medical Society: Honoraria; Merck: Honoraria; Naveris: Honoraria; Nextech: Honoraria; Rain: Honoraria; Regeneron: Honoraria; Replimune: Honoraria; SIRPant: Honoraria; Surface Oncology: Honoraria. Romee:Inndura: Consultancy; Biohaven: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal